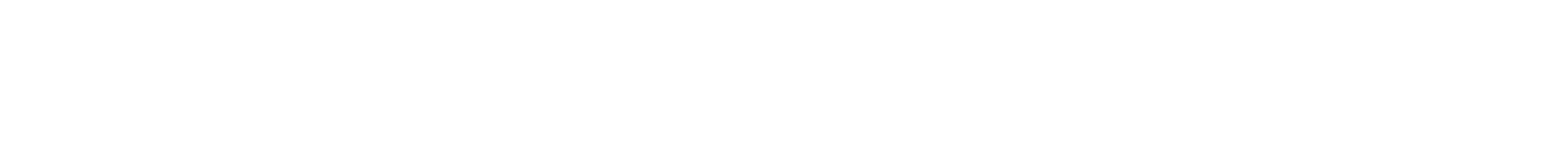Columbia Engineers and Clinicians First to Build a Functional Vascularized Lung Scaffold
New approach to bioengineering lungs selectively treats epithelium that lines the lung airway, while preserving lung vasculature; this transformative technique could lead to improved care of patients with lung disease
End-stage lung disease is the third leading cause of death worldwide, accounting for 400,000 deaths per year in the United States alone. Researchers exploring new ways to both promote lung repair and increase the number of available donor lungs have been challenged by the organ’s extreme complexity—the lung has more than 40 different cell types residing in its matrix and the total surface area between the airway and the vasculature is the size of a tennis court. Efforts to bioengineer functional lungs from fully decellularized or synthetic scaffolds that lack functional vasculature have been largely unsuccessful until now.
A Columbia Engineering team led by Gordana Vunjak-Novakovic, University Professor and the Mikati Foundation Professor at Columbia Engineering and professor of medical sciences (in Medicine) at Columbia University, and N. Valerio Dorrello, assistant professor of pediatrics at Columbia University Medical Center, is the first to successfully bioengineer a functional lung with perfusable and healthy vasculature in an ex vivo rodent lung. Their new approach (DOI 10.1126/sciadv.1700521), which allows the removal of the pulmonary epithelium while maintaining the viability and function of the vascular network and the lung matrix, is published today in Science Advances.
In the past seven years, several research groups have been able to fully decellularize lungs to build scaffolds for lung bioengineering with the proper architecture and stiffness that can be repopulated by newly introduced cells. To rebuild a functional lung from this cell-free scaffold, researchers need to regenerate both the epithelial surfaces in the airway and the endothelial lining of the vasculature. An intact vascular network—missing in these scaffolds—is critical not only for maintaining the blood-gas barrier and allowing for proper graft function, but also for supporting the cells introduced to regenerate the lung. This has proved to be a daunting challenge.
“We developed a radically new approach to bioengineering of the lung,” says Vunjak-Novakovic, a pioneer in tissue engineering who directs the Laboratory for Stem Cells and Tissue Engineering. “We reasoned that an ideal lung scaffold would need to have perfusable and healthy vasculature, and so we developed a method that maintains fully functional lung vasculature while we remove defective epithelial lining of the airways and replace it with healthy therapeutic cells. This ability to selectively treat the pulmonary epithelium is important, as most lung conditions are diseases of the epithelium.”
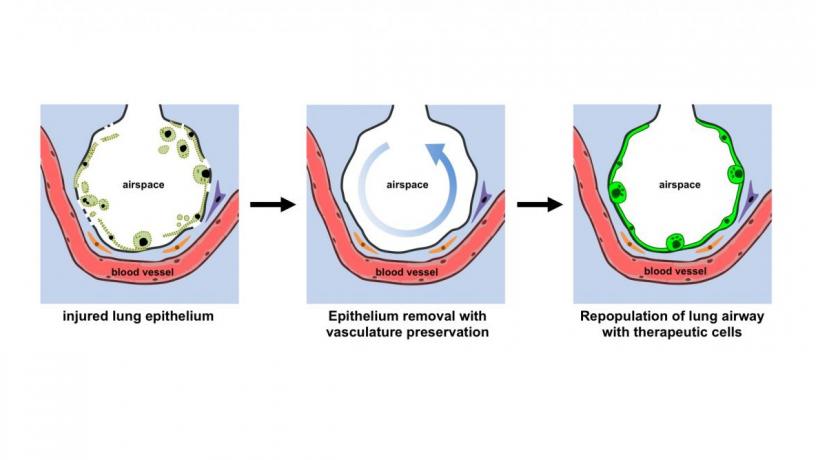
Earlier techniques for decellularization of the entire lung were designed to remove all cells from the lung, generating a cell-free lung scaffold. The Columbia team has now demonstrated a transformative approach to obtaining functional vascularized lung grafts. They developed an airway-specific method to remove the pulmonary epithelium while preserving the lung vasculature, matrix and other supporting cell types such as fibroblasts, myocytes, chondrocytes, and pericytes. Following lung cannulation in a rodent lung, they ventilated the lungs and perfused them on an ex vivo lung perfusion system (EVLP), similar to the extracorporeal membrane oxygenation (ECMO) system utilized to support patients with cardiovascular and respiratory failure. They then delivered intratracheally a mild detergent solution to an isolated single lung to remove epithelial cells and protected the vasculature by circulating a perfusate containing electrolytes and energy substrates. The lung scaffold maintained the bronchial and vascular architecture, and supported the attachment and growth of human adult and stem cell–derived pulmonary cells in this ex vivo bioreactor.
“Every day, I see children in intensive care with severe lung disease who depend on mechanical ventilation support. The approach we established could lead to entirely new treatment modalities for these patients, designed to regenerate lungs by treating their injured epithelium“ says Dorrello, the lead author of the study.
Preserved vasculature in the treated lung being perfused with microspheres
“Strategies aimed at increasing the number of transplantable lungs would have an immediate and profound impact,” says Matthew Bacchetta, associate professor of surgery, Columbia University Medical Center, and a co-author of the paper. “Lung transplantation is the only definitive treatment we have for patients with end-stage lung disease, but there is a severe shortage of donor organs—only 20 percent of potential donor lungs are acceptable for transplantation and this leads to many potentially avoidable deaths on the waiting list. In addition, early intervention with gene or cell therapy may offer even greater benefits by promoting lung repair and regeneration, thus slowing the progression of disease and ultimately avoiding the need for transplantation. This approach could also serve as a means to make the donor lung more resilient and durable for transplantation. As a lung transplant surgeon, I am very excited about the great potential of our technique.”
The study has been supported by a new $8.2M seven-year grant from the National Institutes of Health that focuses on modeling, pathogenesis, and treatment of idiopathic pulmonary fibrosis. The team is already using their controllable model of lung regeneration to treat human lungs ex vivo, and for further study of lung development and repair in the context of the whole organ. They are developing new modalities for imaging-guided cell removal and delivery of therapeutic cells or agents for clinical applications. They are also refining targeted, localized, and selective treatment of the defective pulmonary epithelium to address the clinical needs for specific lung diseases.
“This is a major step forward in bioengineering lungs,” Vunjak-Novakovic says. “The creation of de-epithelialized whole lungs with functional vasculature may open new frontiers in lung bioengineering and regenerative medicine. This project could not have been successfully completed without the interdisciplinary effort that made us step out from our zone of comfort, and try novel approaches in collaboration with bioengineers, clinical scientists, and stem cell scientists.”
Tissue Engineering with Gordana Vunjak-Novakovic